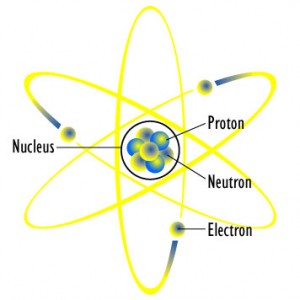
The particles found inside an atom are the proton, neutron and electron. The two particles found in the nucleus are protons and neutrons and these can both be referred to as nucleons.
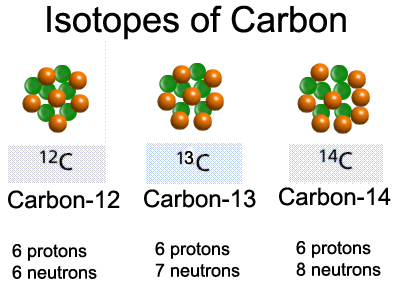
Some atoms have two or more isotopes. This means that different atoms contain different numbers of neutrons and so have different masses. When writing nuclear symbols the mass number is shown on the top left. When writing the name of the isotope using words the mass number is given after the word as shown in the image below.
Why is this relevant to radioactivity? Well the structure of the nucleus is sometimes unstable depending on the arrangement of protons and neutrons. In the carbon example above Carbon-12 and Carbon-13 have stable nuclei but Carbon-14 is unstable. This means Carbon-14 can undergo radioactive decay. An unstable and hence radioactive nucleus such as this is often referred to as a radionuclide.
This link gives a list of common radioisotopes and their uses.
Radioisotopes
The short video at the link below explains what these nucleons (particles of the nucleus) have to do with radioactive decay.
Video: Uranium - Twisting the Dragon's Tail
This link gives a list of common radioisotopes and their uses.
Radioisotopes
The short video at the link below explains what these nucleons (particles of the nucleus) have to do with radioactive decay.
Video: Uranium - Twisting the Dragon's Tail